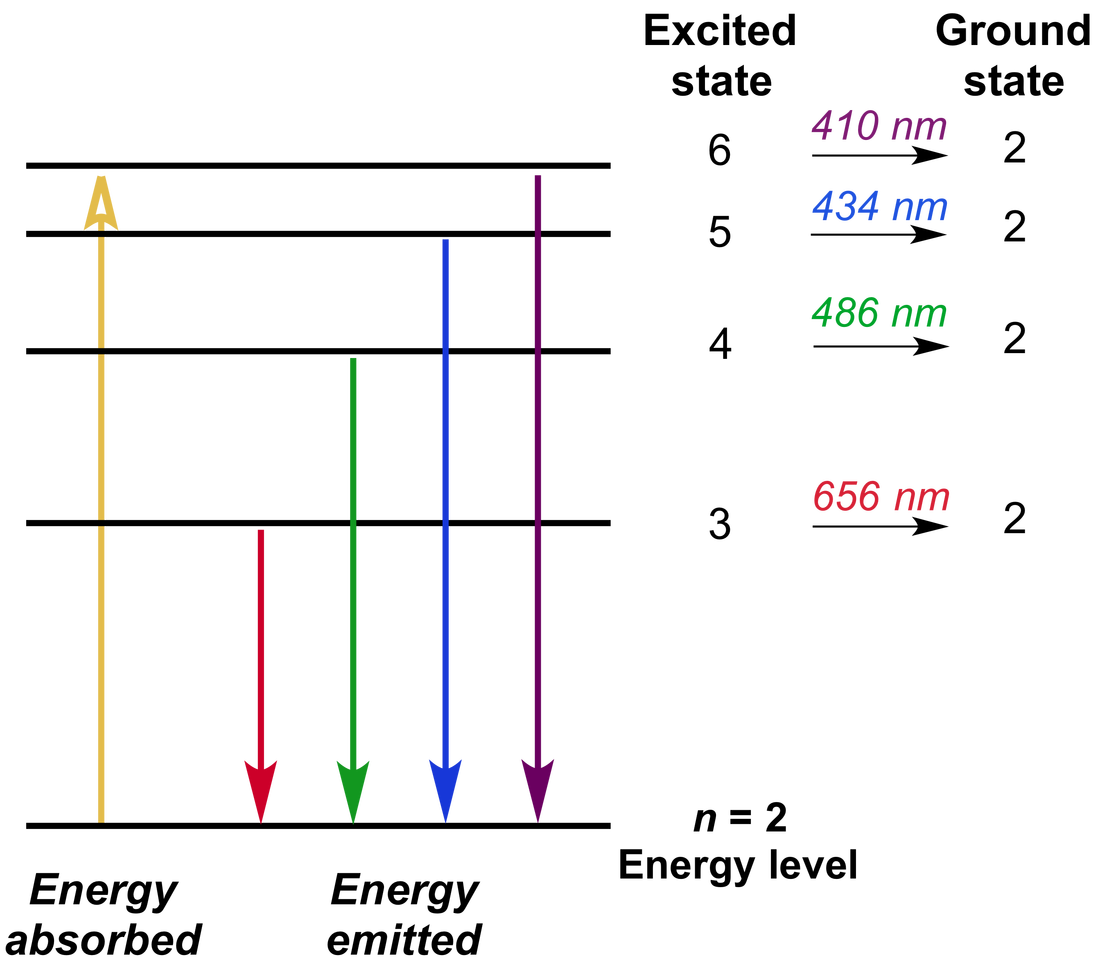
The analytical instruments that exploit the atomic emission consist of the following main parts: (a) The flame (or an oven in other similar techniques). Lithium’s red line owns at least 10 times greater intensity compared to the other ones. Representation of the persistent lines of lithium (shown carmine in flame tests) and potassium (lilac in flame tests). The linear spectra of lithium and potassium are also provided in Figure 2. Each element’s atomic linear spectrum is characteristic. In the case of atoms the spectrum consists of specific frequencies and is therefore called linear. The sum of a substance’s emitted wavelengths constitute its emission spectrum. The whole procedure is summarized in Figure 1. Being ν = c/ λ it is possible to calculate the wavelengths of the emitted radiations that result to be equal to: 590 nm, 330 nm and 285 nm for the transitions: 3p → 3s 4p → 3s and 5 p → 3 s, respectively. The excited electron of the sodium atoms will return to their fundamental energy state emitting a radiation having energy hν equal to the difference in the energy of two levels involved in the transition. However, the produced excited states are not energy stable. As a result, the electron from the 3 s orbital can be found in 3 p, 4 p or 5 p orbitals. But when they are in the flame, they get excited. For example, sodium atoms, in their ground state own the 11Na: 1s 2 2s 2 2p 6 3s 1 configuration. The theoretical backgroundĮmission spectrophotometry methods rely on the emission of light from excited atoms. Thus, in our report, firstly, we describe all the necessary steps in order to construct and calibrate a DIY flame emission spectrophotometer starting from very cheap materials and then we discuss the application of our device in the detection of metal ions in commercially available drug formulations. Such an instrument could provide the opportunity of the introduction of emission spectroscopy in the school lab. 
But none of them discusses the possibility of the construction of a simple DIY device like this one within the framework of the upper secondary chemistry curricula. The theremino documentation (available at ) provides a procedure for constructing a spectrometer (the heart of the AES), while some youtubers have already performed relative tasks ( Electrical Projects, 2018 YouTube account PleWesley, 2016). The advantage of the aforementioned software over the other solutions is that it can easily interpret the signal from a low cost USB camera providing an excellent opportunity for the students to construct themselves their own analytical device ( vide infra). Moreover, some “smart” phone’ apps recognize intensity of certain colors as well (such as Color Grab or ColorMeter for Android phones). Of course, analogous software are already available in expensive cameras. Nevertheless, recently, a company named Theremino (Theremino Group, ) freely distributed a software that analyzes the intensity of the incident light on a camera and provides an intensity vs. The discussion about metals’ emission spectra in most of the cases stops at the performance in the school lab of the well known flame tests ( Shakhashiri, 2011). Unfortunately, this fundamental technique and its underlying science is usually absent from the secondary education chemistry curricula. In the case of quantitative analysis, the intensity of the radiation emitted is related to the amount of the analyte present in the sample. Furthermore, the study of the structure of our galaxy and the study of the expansion of our Universe is made possible through an analogous technique ( Freedman & Madore, 2010). .jpg)
This method is based on the fact that the wavelengths of the radiation emitted by an excited sample are characteristic of the atoms present therein. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.One of the first methods developed in order to achieve the detection of metals in various samples along with their quantitative determination was atomic emission spectrophotometry (AES) ( Skoog, West, Holler, & Crouch, 2014 Strobel & Heineman, 1989). Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
